
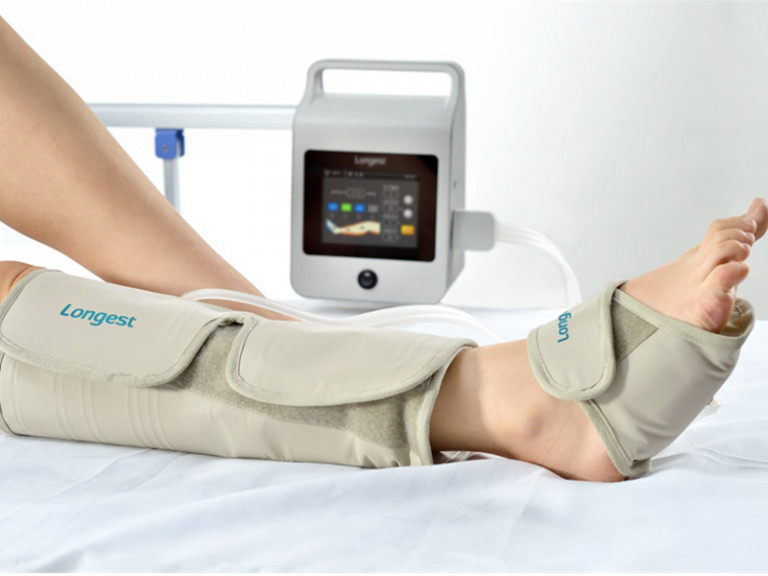
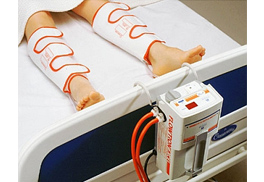
Segmental lymphedema pumps, previously noncovered, are now covered as DME if necessary criteria are met. The only time that a segmented, calibrated gradient pneumatic compression device (HCPCs code E0652) would be covered is when the individual has unique characteristics that prevent them from receiving satisfactory pneumatic compression treatment using a nonsegmented device in conjunction with a segmented appliance or a segmented compression device without manual control of pressure in each chamber.Ġ9/1986 - Added section to place lymphedema pumps and accompanying information together. The clinical response includes the change in pre-treatment measurements, ability to tolerate the treatment session and parameters, and ability of the patient (or caregiver) to apply the device for continued use in the home.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
